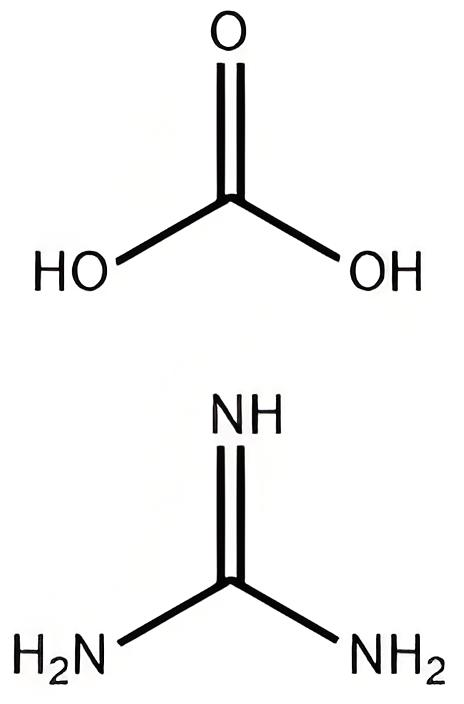
|
Uses
|
Flame Retardant Mechanism:
Endothermic Decomposition: Decomposes at high temperatures to release non-combustible gases (such as CO₂ and NH₃), diluting oxygen concentration and lowering combustion temperature.
Char Catalysis: Promotes the formation of a dense char layer on the material surface, insulating it from heat and oxygen.
Synergistic Effect: When combined with phosphorus or antimony flame retardants, it significantly improves flame retardancy.
Applications:
Cellulose Materials: Suitable for use in wood, cardboard, and other applications, due to its low hygroscopicity and minimal decorative effects, making it suitable for high-end decorative materials.
Intumescent Fire Retardant Coatings: As a foaming component, it can be combined with melamine and other materials to enhance char density.
Engineering Plastics such as Nylon: Requires use in conjunction with other flame retardants (such as MCA) to achieve UL94 V-0 rating.
Advantages and Limitations:
Advantages: Low toxicity, halogen-free, and environmentally friendly;
Minor impact on material mechanical properties (such as tear strength);
Limitations:
When used alone, flame retardant efficiency is limited and requires compounding with other flame retardants;
May generate ammonia at high temperatures, requiring smoke emission control;
|